How to write an orbital diagram Top notch tips about how to draw orbital diagrams Orbital diagrams and electron configuration
How To Write An Orbital Diagram
Localized bonding and hybrid atomic orbitals Describe the shape of a p orbital. Orbital orbitals atomic chemistry shapes energy probability tutorial basics crash academy
Atomic orbitals explained
Orbital molecular diagram chemistry theory draw two mo energy bond o2 order electrons shown ca diagrams oxygen bonding unpaired labeledOrbital energy diagram 8.4: molecular orbital theoryElectron orbitals electrons quantum numbers chemistry electronic structure atoms introductory orbital model figure atomic number arrangement libretexts ball chapter not.
Electron configurationsBromine orbital diagram Orbital diagrams — overview & examples41 what does each box in an orbital diagram represent.

14+ hbr mo diagram
Orbital orbitals chemistry meaning chem electronOrbital diagrams Blank orbital diagram3.7: electron arrangement- the quantum model.
Orbital molecular theory diagram libretexts chemistry bond energy chem electrons order unpaired bonding deki revision apiBonding molecular between antibonding orbital orbitals mo bonds theory difference pi covalent diagram ethylene energy electron multiple chemistry polyatomic anti What is the electron configuration orbital diagram class chemistry cbseOrbitals hybridization hybrid chemistry orbital sp3 atomic hybridized three atoms sp each bond four valence equivalent blue red produces bonding.

Electronic pairing structure orbital diagrams chemistry quantum diagram notation spin box electrons electron orbitals energy first spins configurations boxes level
Figure8.6.electron orbitalsOrbital electron diagrams configuration practice chemistry problems basic Hybrid atomic orbitalsOrbital diagrams.
Electron configuration orbital table periodic order chart electrons config configurations per shells following use just chemOrbital electron diagrams element monahan difference Atomic orbitals explainedDistribution of electrons in different orbits [with examples].

Molecular orbital theory
Orbitals hybridization hybrid atomic bonding chemistry molecular theory sp localized electronic bond geometry orbital atom hybridized example using section structure15+ ground state orbital diagram How to write orbital filling diagrams for atoms: examples & practicePauli exclusion principle.
Orbital electron diagrams configuration diagram 2s atom configurations 3s 2p 1s potassium ppt powerpoint presentation 3p slideserve thereOrbital molecular he2 be2 o2 bonding paramagnetic diamagnetic orbitals electrons diatomic molecules chem unpaired labeled antibonding inorganic mixing nitrogen atoms N2o molecular orbital diagramPolyatomic systems with multiple bonds.
[diagram] electron configuration and orbital diagram answers
Molecular orbital theoryOrbital diagrams Orbitals, the basics: atomic orbital tutorial — probability, shapesDraw the molecular orbital diagram for ne2 and determine if the bond.
Electron orbitals quantum chemistry electrons numbers electronic structure model atoms introductory orbital number figure arrangement atomic chem each energy libretextsOrbits electrons electron shells capacity order nucleus teachoo maximum .


Orbital Diagrams - Chemistry Steps

Orbital - Definition, Diagram, Meaning - Study Chemistry

Hybrid Atomic Orbitals | Chemistry I

PPT - Orbital Diagrams and Electron Configuration PowerPoint

Molecular Orbital Theory | Chemistry for Majors
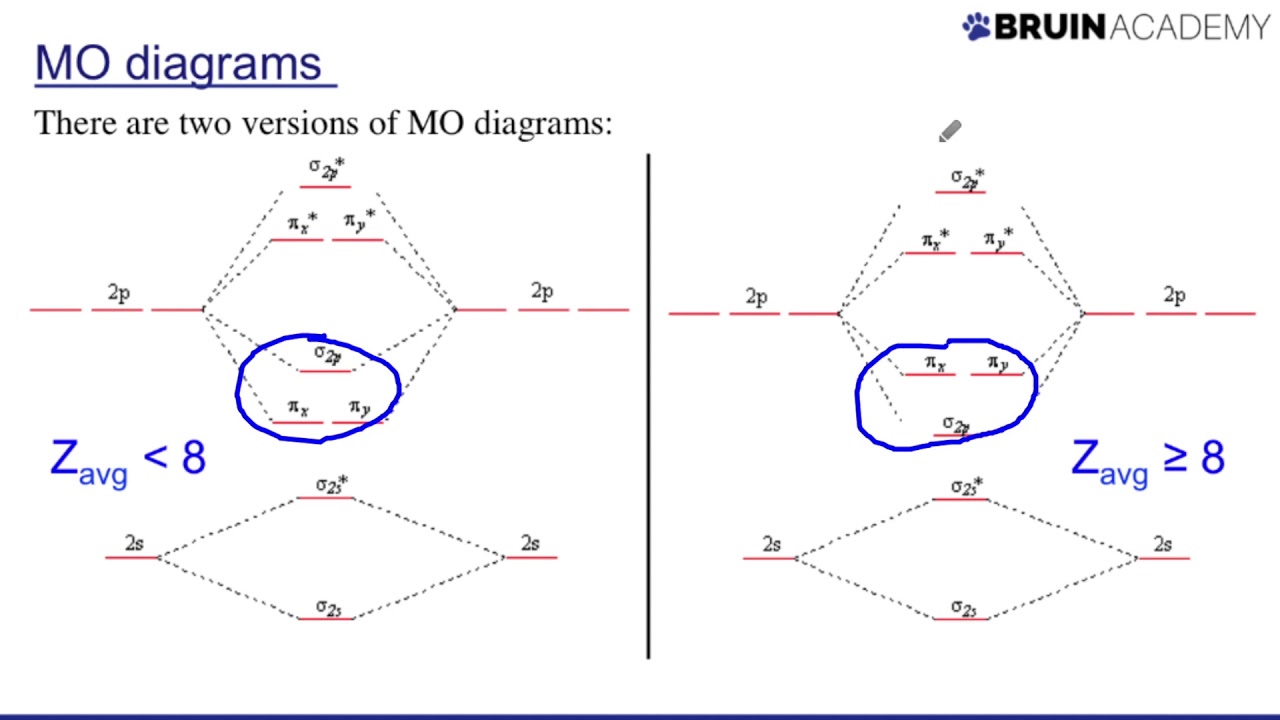
14+ hbr mo diagram

Describe the Shape of a P Orbital.